We are applying our expertise in genomic profiling to identify elusive cancer-related genetic changes and new biomarker targets across many cancer types. Our advanced cancer genome analysis helps researchers and biopharmaceutical partners accelerate new precision medicine development.
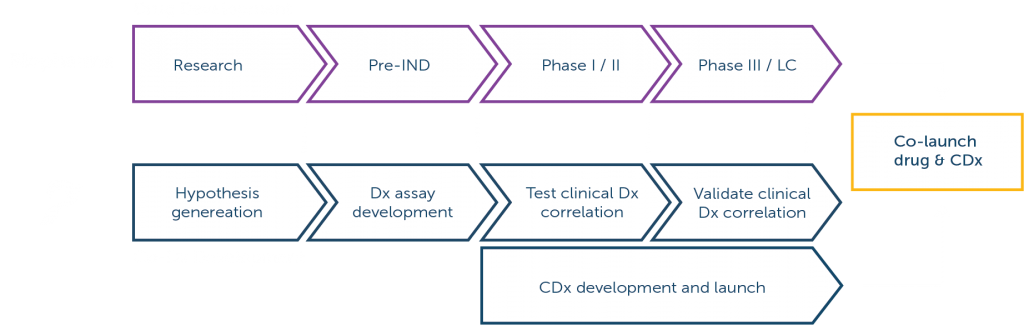
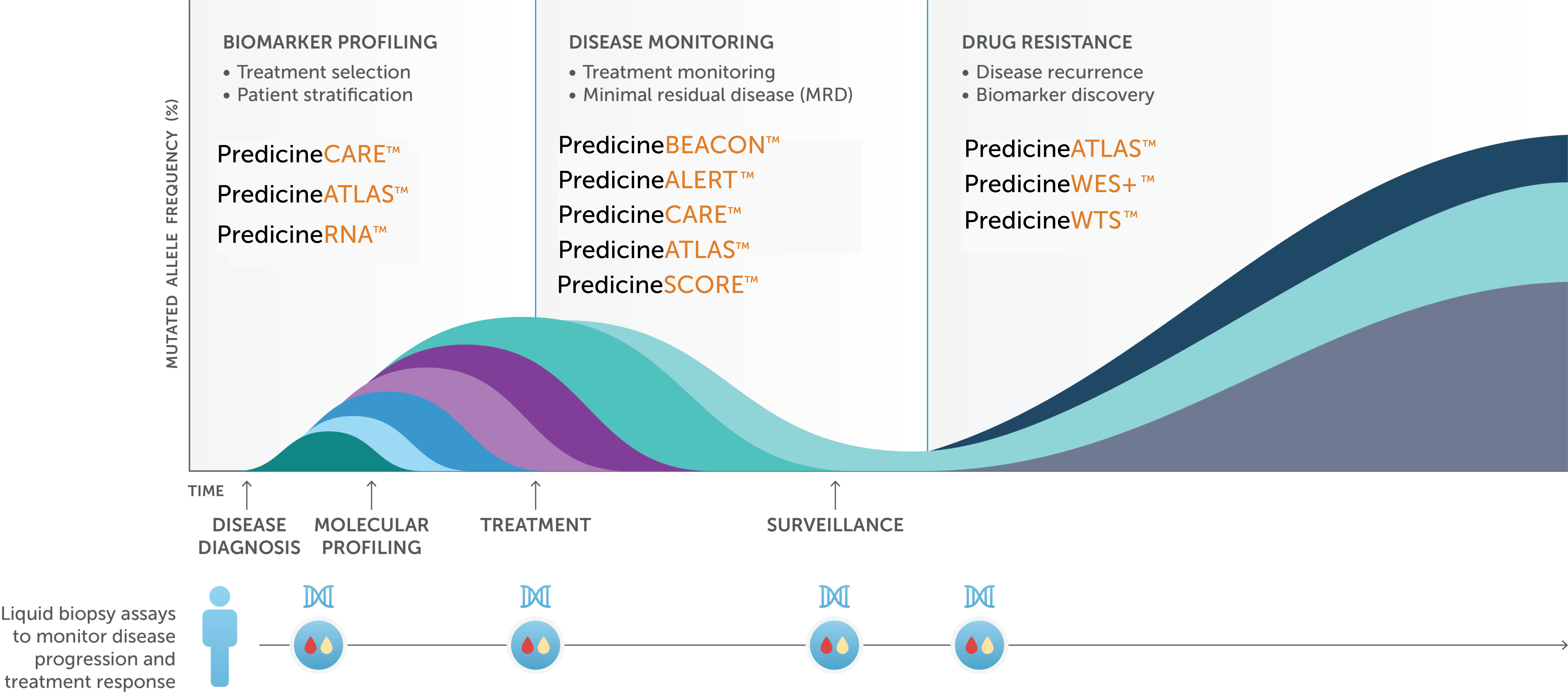
We offer a portfolio of comprehensive genomic profiling assays for precision medicine development.
| ALL SOLID TUMORS | HEMATOLOGIC MALIGNANCIES | ||||
|---|---|---|---|---|---|
| COMPARE | PredicineATLAS™ | PredicineCARE™ | PredicineRNA™ | PredicineCARE™ Ultra | PredicineHEME™ |
| OVERVIEW | Broadest gene coverage | Pan-cancer panel (includes guideline-recommended genes) | Panel for known or novel gene fusion / splice variant | Ultra-Sensitive cfDNA panel | Panel for hematologic malignancies |
| NUMBER OF GENES | 600 (DNA) | 200 (DNA) | 88 (RNA) | 158 (DNA) | 259 (DNA) |
| SPECIMEN TYPE | Blood Urine Tissue | Blood Urine Tissue | Blood Tissue | Blood Urine | Blood Bone marrow aspirate |
| TURNAROUND TIME | Plasma: 6 Days Tissue: 10 Days | Plasma: 6 Days Tissue: 10 Days | 14 Days | 6 Days | 6 Days |
| TMB | ✓ | ✓ | — | — | — |
| MSI | ✓ | ✓ | — | — | — |
| MMR | ✓ | ✓ | — | — | — |
| HRD | ✓ | ✓ | — | — | — |
| REPORT FEATURES | SNVs INDELs CNA CNL Rearrangements | SNVs INDELs CNA CNL Rearrangements | RNA fusions RNA splice variants | SNVs INDELs CNA CNL Rearrangements | SNVs INDELs CNA Rearrangements |
| DEPTH | 20,000x | 20,000x | — | 100,000x | 20,000x |
| VERSIONS | CLIA RUO | CLIA RUO | RUO | CLIA RUO | CLIA RUO |
We provide harmonized assays supported by unified quality management systems and workflow in our network of CLIA-certified and CAP-accredited facilities in U.S. and China to support cross-border clinical studies.
Ready to streamline your precision oncology clinical research programs? Submit your information to speak to a team member.