Analyze cell-free circulating DNA and RNA in blood and urine
Unlock complex genomic data for precision medicine

Decipher cancer genome to accelerate oncology drug development

Simultaneous profiling of DNA and RNA from a tube of blood for more insights
LEARN MOREMore robust variant detection in urine than in blood in bladder cancer
LEARN MORE
Integrated molecular and contextual tissue biomarker solution
LEARN MORERobust and sensitive ctDNA variant profiling from as little as 0.4mL of plasma
LEARN MOREPredicine’s proprietary GeneRADAR next generation liquid biopsy platform measures cancer-associated genomic blueprint (DNA) and functional biology (RNA) in a single tube of blood to provide more accurate and in-depth insights.
Predicine introduces urine-based liquid biopsy to broaden cancer patients’ access to genomic profiling. Urine-based cfDNA can be more robust in variant detection than blood-based cfDNA in genitourinary cancers.



“Liquid biopsy is the future of precision medicine. Our work with Predicine’s liquid biopsy platform is very encouraging for the field of liquid biopsy and supports its clinical application for patient care.”
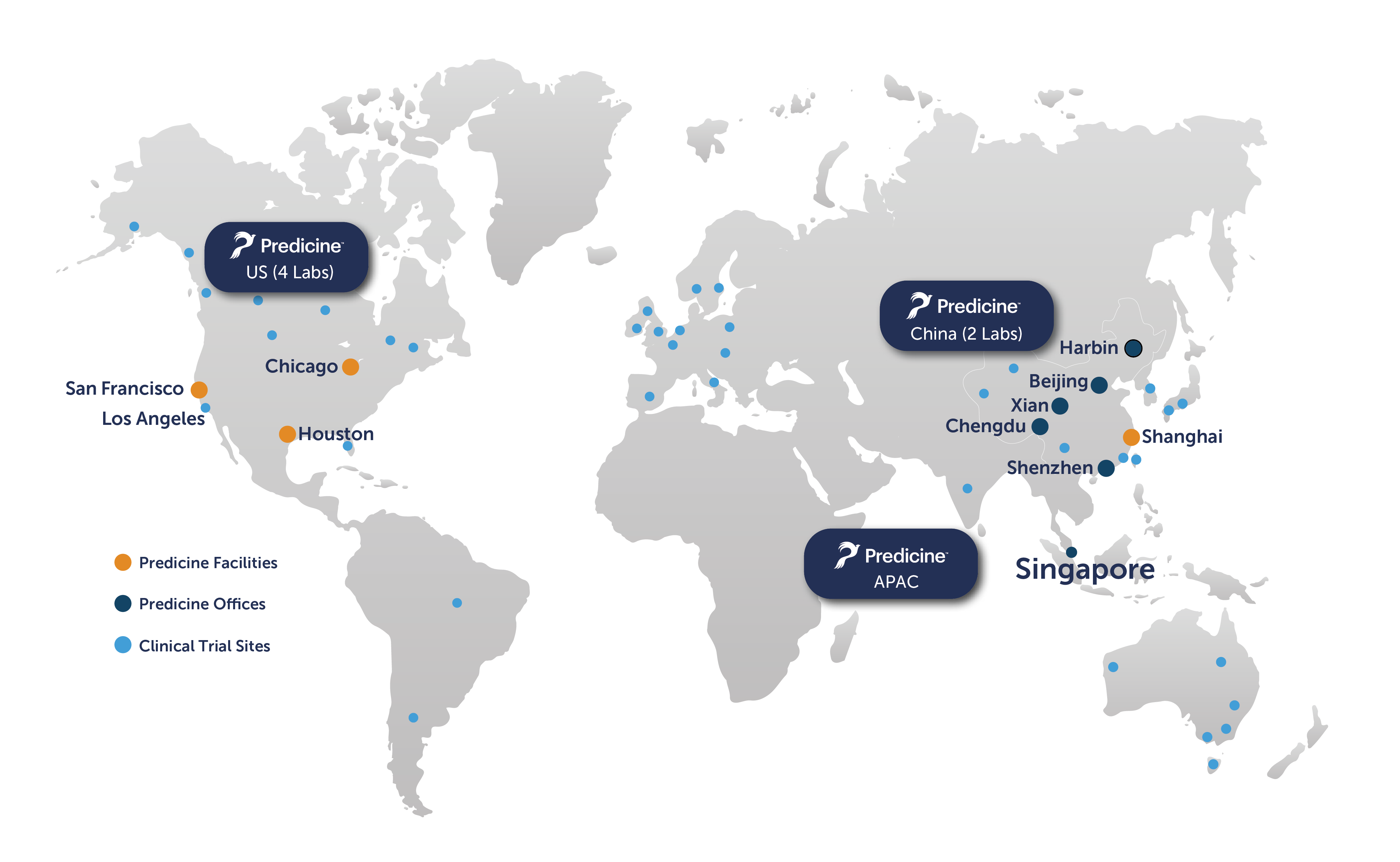
Through our network of CLIA/CAP certified facilities in California and Shanghai, Predicine partners with leading biopharmaceutical companies to support biomarker-driven clinical trials.
Ready to streamline your precision oncology clinical research programs? Submit your info via the form below and we will respond within 24 hours.